Client Communication
2024
Herpes Simplex Virus 1 & 2 Nucleic Acid Amplification Testing
Effective immediately, order code 4044 HERPES SIMPLEX CULTURE+TYPING has been discontinued by our reference laboratory. For qualitative detection and differentiation of Herpes Simplex Virus 1 and 2 (HSV-1 and -2) the replacement order code for this test is order code 5367 HSV 1/2 BY PCR.
PCR has been shown to be more sensitive than culture and is the preferred diagnostic testing method. NAAT methods are the preferred method, with PCR as the most widely used NAAT method
Methodology: Real-Time Polymerase Chain Reaction (RT-PCR)
Preferred Specimen Requirements: VIRUS TRANSPORT MEDIA (VTM)
Acceptable Specimen Requirements:
- ESWAB (WHITE)
- THINPREP VIAL
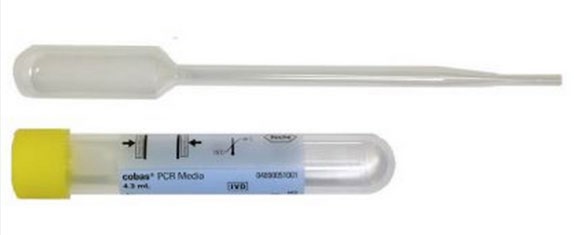
- ROCHE COBAS PCR MEDIA
- SERUM FROM SERUM SEPARATOR TUBE
- EDTA WHOLE BLOOD
- EDTA PLASMA
- BODY FLUID (AMNIOTIC)
- LAVAGE, CSF, OCULAR
- BRONCHOALVEOLAR
Transport Temperature: Refrigerate
Please contact your East Side Clinical Laboratory Sales Representative with any questions
Transition to New Stool Collection Containers for Stool Culture, Gastrointestinal Panel-PCR, and Ova & Parasite
In an effort to better adhere to clinical literature and preserve stool specimens for testing, East Side Clinical Laboratory will be distributing new collection containers for stool testing.
The transition to these new containers will improve the recovery and success of the following tests: Stool Culture, Gastrointestinal Pathogen Panel, and Ova & Parasite Screening.
The laboratory has determined:
- Copan FecalSwab (Cary-Blair) and Apacor ParaSep are the best devices for ease, and will provide clients and patients a better option for collection.
- Current clinical practice for stool culture recommends Cary-Blair (FecalSwab) is used to improve recovery of isolates for culture. Organisms such as Shigella can become non-viable if raw stool is not placed into preservative within two hours of collection. FecalSwab will also be instrumental in the preservation of stool for molecular testing of the Gastrointestinal Panel.
- It is also imperative that stool for Ova & Parasite Screening be immediately placed into appropriate preservative ParaSep for adequate stability of organisms. Raw stool specimens should be placed into the preservative within three hours of collection for optimal recovery and preservation.
- The benefit of this transition will provide a better streamline and maintain suitable stability. This will also provide the specimen a longer stability period once the specimen is placed into preservative.
This transition will include the following tests and collection devices:
| Collection Device | 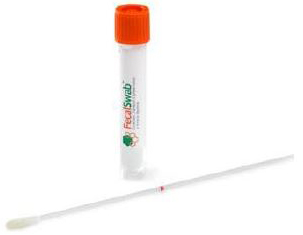 |
 |
| Testing For | Stool Culture Gastrointestinal Panel, PCR |
Ova & Parasite |
| Transfer Time | 2 hours of collection | 3 hours of collection |
| Specimen Stability | 48 hours | 6 months |
| FecalSwab | ParaSep |
|---|
Distribution
Please contact your local ESCL facility for supplies. There will be no charge in the ordering or distribution process.
NOTE: During the transition, ESCL will continue to accept stool with our current practice until fully moved over to the Copan FecalSwab and Apacor ParaSep collection devices.
Please contact your East Side Clinical Laboratory Sales Representative with any questions.
2023
Service Disruptions
Effective December 12, 2023, East Side Clinical Laboratory’s logistics, supply distribution, and personnel allocation will be impacted by the partial closure of the Washington Bridge (RT. 195).
Our headquarters and primary laboratory, located in East Providence, will be making changes in real-time to accommodate for the changing traffic and detour patterns caused by the current issues on the Washington Bridge. We do expect delays in our pickups and we are doing the best we can to find alternate routes.
We kindly ask for your patience during these challenging times and further commit to our excellent services known to our patients, providers, and partners.
Thank you!
Please contact your East Side Clinical Laboratory Account Service Representative with any questions.
2023 Annual Notice to Physicians and Non-Physician Practitioners (NPP)
East Side Clinical Laboratory (ESCL) is providing this notice as part of our compliance program in accordance with OIG recommendations to clinical laboratories.
Periodically, ESCL advises its physicians and clients about program changes and information related to federally‐funded healthcare programs that affect both the provider and laboratory. Please note that these notices are intended to help both the provider/client and the laboratory comply with these regulations and mitigate the risks for all parties.
When ordering tests for which Medicare reimbursement will be sought, the following regulations apply:
Licensed Physicians and Non‐Physician Practitioners (NPP)
A clinical laboratory may only bill Medicare and Medicaid for testing ordered by a licensed provider authorized by law to order laboratory tests. If your license has been revoked or suspended, please immediately notify the laboratory. As of 2014, Medicare requires individuals referring orders for laboratory services on Medicare beneficiaries to be registered in the Center for Medicare and Medicaid Services’ Provider Enrollment, Chain and Ownership System (PECOS). This enrollment is required of all providers, including physicians and non-physician practitioners (NPP), even if they themselves do not offer services to Medicare beneficiaries. Additional information on PECOS and how to enroll in the system may be viewed at: https://pecos.cms.hhs.gov/providers/index.html
Medical Necessity
Tests that are medically necessary for the diagnosis or treatment of a Medicare patient are covered and may be reimbursed by Medicare. An approved test panel must only be ordered when every test in that panel is medically necessary. If all components of the panel are not medically necessary, you should order individual tests or a panel that contains only the medically necessary tests. Screening or Investigational Use Only tests are generally not covered by Medicare, with some exceptions for Wellness Screens. As a Medicare participating provider, ESCL has a responsibility to make good faith efforts to ensure that all tests requested are performed and billed in a manner consistent with all federal and state laws and regulations. The OIG takes the position that providers authorized by law to order laboratory tests, who knowingly cause a false claim to be submitted to any federally funded program, may be subject to sanctions or remedies available under civil, criminal and administrative law, such as the False Claims Act.
American Medical Association (AMA) Organ or Disease‐Oriented Panels
Authorized ordering providers should only order AMA defined Organ or Disease‐Oriented Panels when all components are medically necessary. The AMA panels were developed for coding purposes only and should not be interpreted as clinical parameters.
Note: The AMA panels and their components are listed in an attachment to this notice.
Custom Profiles
A custom profile is a specific group of commonly ordered tests, or panels which have not been defined by the AMA or CMS, which are medically necessary in treating a condition. Custom panels are established as defined by the requesting Healthcare Provider (HCP) and may be used only by that HCP. An ESCL Laboratory Director will be responsible for validating custom profiles by reviewing a signed Physician Acknowledgement annually from each client for each custom profile utilized in the treatment of his/her patients.
The OIG has noted that such profiles present the danger that, by defaulting to such a profile, the physician may in a particular case be ordering one or more medically unnecessary tests contained in the profile. The OIG takes the position that ordering unnecessary tests within a profile, even when done without a specific intent to defraud, may subject the ordering physician to civil penalties. If modification to custom profile is warranted, the requesting physician is advised to contact his/her Client Support Coordinator (CSC) to assist with adding, updating or deleting.
Diagnosis Information
Section 4317 of the Balanced Budget Act of 1997 requires the authorized ordering provider to submit diagnosis information on the laboratory order for submission of a Medicare insurance claim. The diagnosis information supplied should accurately describe the patient’s condition on the date of service as documented in the patient’s medical record. If you are ordering a test and cannot yet determine a diagnosis, you must provide the appropriate ICD-10 code that describes the patient’s signs and symptoms. Laboratories are prohibited from using diagnostic information provided by the physician from earlier dates of service (except for certain standing orders, as described below), and from inserting diagnosis codes based on their own evaluation of the patient’s probable or most likely diagnosis, or from speaking with a patient.
Providers’ offices will be contacted by ESCL in the event that this required information is omitted on any laboratory order, and this communication may occur via telephone call or fax. Additions, Revisions, and Deletions for ICD‐10‐CM codes which became effective on October 1, 2023 may be viewed at: https://www.cms.gov/medicare/coding-billing/icd-10-codes/2018-icd-10-cm-gem
2023 Medicare Clinical Diagnostic Laboratory Tests Payment System
Effective January 1, 2018, the Clinical Laboratory Fee Schedule (CLFS) reimbursement rates were revised to be based on the weighted median private payor rates as required by the Protecting Access to Medicare Act (PAMA) of 2014. Co‐payments and deductibles do not apply to services paid under the Medicare clinical laboratory fee schedule. Also, Medicaid reimbursement will be equal to, or lesser than Medicare reimbursement.
The 2023 Medicare Clinical Laboratory Fee schedule may be viewed and downloaded at: https://www.cms.gov/medicaremedicare-fee-service-paymentclinicallabfeeschedclinical-laboratory-fee-schedule-files/22clabq4 for the fee schedule listing.
Medicare Laboratory Local Coverage Determinations (LCDs) and National Coverage Determinations (NCDs)
Coverage determination policies define the medical conditions through the inclusion of a list of ICD‐10 (diagnosis) codes for which tests are covered or reimbursed by Medicare. HIPAA regulations require ICD‐10 code(s) to be present on each claim filed. These codes must also be documented in the patient’s medical record. Property and casualty, auto and workers' compensation insurance are not covered entities and are therefore not required to comply with HIPAA. All laboratory NCDs may be viewed at: https://www.cms.gov/Medicare/Coverage/CoverageGenInfo/LabNCDsICD10.
To search the Medicare coverage database: https://www.cms.gov/medicare-coverage-database/new-search/search.aspx . The Centers for Medicare and Medicaid Services has authorized NGS Medicare, as a jurisdiction Medicare Part B carrier, to develop Local Coverage Determinations (LCD). These guidelines may supplement or be in addition to the National Coverage Determinations and give direction for medical necessity on selected tests. The LCDs may be viewed at: https://www.ngsmedicare.com/medical-policies?lob=96664&state=97133&rgion=93623
Advance Beneficiary Notice of Non-coverage (ABN)
Not all laboratory services are covered by Medicare. For statutorily excluded services, ESCL may bill Medicare patients directly. For certain other laboratory tests, an Advance Beneficiary Notice of Non-coverage (ABN) is used to document that the patient has been made aware that Medicare may not pay for service(s) and has agreed to pay the laboratory in the event payment is denied by Medicare. A separate ABN must be used/completed for each specimen collection encounter. ESCL will provide ABN forms to clients at their request. Client‐collected ABNs must accompany the order/requisition when samples are submitted to the laboratory.
ESCL be responsible for collecting the ABN from the patient when there is a face‐to‐face interaction between the patient and a laboratory staff member. Please check the expiration date located in the lower left hand corner of the ABN, to assure the most current form (Exp. 01/31/2026) is being utilized and completed, when an ABN is needed for a Medicare beneficiary. Information about ABNs may be viewed at: https://www.cms.gov/Medicare/Medicare-General-Information/BNI/ABN
Billing Information
ESCL requires the following information to enable our Billing Department to bill Medicare, Medicaid or other commercial insurance. The ESCL requisition contains adequate, clearly labeled spaces to provide this information:
- Date of Order
- Patient’s full name,
- Patient’s current complete address, city, state and zip code,
- Date of birth and gender,
- Patient’s insurance company name, ID number including prefix/suffix, if applicable.
- Ordering/referring Provider’s name and NPI number,
- Valid ICD‐10‐CM diagnosis code(s) for each test, not just Medicare Limited Coverage Tests, and
- A valid ABN, when mandated by Medicare NCD/LCD policy.
Provider Signatures
The revised Medicare Learning Network Fact Sheet (MLN) now requires the ordering provider’s handwritten or electronic signature to show proof of an authenticated medical record supporting the provider’s intent to order specific tests. In order to comply with this requirement, ESCL will contact the provider to retrieve such authentication prior to providing laboratory services to patients. This information may be viewed at https://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/Downloads/ProviderComplianceLabServices-Fact-Sheet-ICN909221.pdf
Reflex Testing
Reflex testing occurs when initial test results are positive or outside normal parameters and indicate that a second related test is medically appropriate for patient care because some results trigger multiple tests (cascade testing). Please see the laboratory test directory for tests that may require reflex testing. Whenever you order an initial test subject to a reflex test, you must consider whether the reflex test is, in your judgment, medically necessary for that particular patient. If you consider the reflex test unnecessary, order the initial test without the reflex test on the requisition form.
Preventive/Screening Services
Statutorily, Medicare does NOT cover any lab testing for routine and/or screening purposes. However, Medicare does cover some Preventive lab tests (PSA, Glucose, Lipids, etc.) if ordered as required by Medicare. For Preventive benefit information including test names, CPT codes, required ICD-10 codes and frequency limitations, please reference: https://www.cms.gov/Medicare/Prevention/PrevntionGenInfo
Medicare Part B National Correct Coding Initiative (NCCI) Edits
The Medicare NCCI was implemented to promote national correct coding methodologies and to control improper coding leading to inappropriate payment. Information on the NCCI edits may be found at: https://www.cms.gov/Medicare/Coding/NationalCorrectCodInitEd/index.html.
Prohibited Referrals and Inducements
It is the policy of ESCL to comply with both the Physician Self-Referral Law (Stark) and the Anti-Kickback Statute. If a financial relationship exists between a physician (or their immediate family member) and a laboratory, the Stark Law prohibits the physician from referring Medicare beneficiaries to the laboratory, and the laboratory cannot bill Medicare for any services referred by the physician unless the financial relationship between the parties falls into one of the law’s exceptions. The Anti-Kickback Statute prohibits the knowing or willful offer, payment, solicitation, or receipt of remuneration in order to induce business reimbursed under the Medicare or Medicaid programs. Any form of kickback or inducement to secure Medicare or Medicaid referrals is strictly prohibited.
Client Services
Client Services representatives are available at telephone number (800)-479-5227 for Client Services. Please take a few minutes to review this information with your staff. We value your business and appreciate the opportunity to serve your laboratory needs in conjunction with these initiatives. If there are further questions regarding this information, please contact your Account Executive or Client Service Coordinator.
Reporting
Should you observe or suspect any violations as mentioned under Prohibited Referrals and Inducement section of this notification, please alert East Side Clinical Laboratory Compliance Department:
Compliance Department
250 Miller Place
Hicksville, NY 11801
Phone: 800.782.0282 x1658
Email: Compliance@sunriselab.com
Laboratory/Clinical Questions
Laboratory Director
Walther Pfeifer, MD
Phone: 401-455-8409
Email: wpfeifer@esclab.com
Change in Testing: Troponin T
Effective December 4, 2023:
East Side Clinical Laboratory is pleased to announce the conversion of troponin testing from the current troponin T assay to the enhanced generation 5 cardiac troponin T (Roche, cTnT GEN5) for the comprehensive evaluation of AMI. The new assay is a high-sensitivity cardiac troponin that enables more sensitive and precise detection at low troponin concentrations.
Troponin T (TnT) is a component of the contractile apparatus of the striated musculature. Although the function of TnT is the same in all striated muscles, the cardiac Isoform of TnT originating exclusively from the myocardium (cardiac TnT, molecular weight 39.7 kDa) clearly differs from skeletal muscle TnT. As a result of its high tissue-specificity, cardiac troponin T (cTnT) is a cardio-specific, highly sensitive marker for myocardial damage.
Rapid, precise, and accurate detection of clinical, imaging, electrophysiological, and biomarker findings are the foundation for the diagnosis of acute myocardial Infraction (AMI). Although current cardiac-specific assays are effective, they are often limited by decreased sensitivity and impression.
The Universal Definition of Myocardial Infraction (UDMI) recommends as an “optimal precision” the coefficient of variation (CV) at the 99th percentile upper reference limit to be less than 10%. The Roche Generation 5 Cardiac Troponin T meets the UDMI criteria for “guideline acceptable” precision and the International Federation of Clinical Chemistry (IFCC) definition of a high sensitivity assay.
Important Changes to Note:
- Units of Measure: Results are currently reported in ug/L. The new troponin 5th generation will be reported in ng/L.
- Only Li-Heparin Plasma Specimens: Test is not FDA approved on serum. Serum specimens will be rejected.
- Sex-Specific Reference Ranges: < 14 ng/L for females and < 22 ng/L for males.
- Critical Value Has Changed: The new critical value for the 5th generation troponin assay will be ≥ 14 ng/L for females and ≥ 22 ng/L for males.
| Troponin T | 8180 |
| Name | Test Code |
|---|
Changes in Reporting:
| Assay | Test Code | Previous Clinical Reporting Range | New Clinical Reporting Range |
| Troponin T | 8180 | 0.010 – 250 ug/L | 6 – 10,000 ng/L |
| Assay | Test Code | Previous Reference Range | New Reference Range |
| Troponin T | 8180 | < 0.010 ug/L |
< 14 ng/L for females < 22 ng/L for males |
| Assay | Test Code | Previous Critical Value | New Critical Value |
| Troponin T | 8180 | ≥ 0.010 ug/L |
≥ 14 ng/L for females ≥ 22 ng/L for males |
| Order-Unit Code and Test Names | 8180 Troponin T |
| Specimen Requirements: | 1 ML LI-HEPARIN PLASMA LI-HEPARIN PLASMA TUBES CONTAINING SEPARATING GEL CAN BE USED. CENTRIFUGE SAMPLE WITHIN 2 HOURS OF COLLECTION. TRANSFER PLASMA TO A PLASTIC TRANSPORT TUBE. CLEARLY LABEL AS HEPARIN PLASMA. WHEN MULTIPLE TESTS ARE ORDERED, SUBMIT SEPARATE TUBE FOR THIS TEST. NOTE: DO NOT COLLECT SAMPLES FROM PATIENTS RECEIVING THERAPY WITH HIGH BIOTIN DOSES (> 5 MG/DAY) UNTIL AT LEAST 8 HOURS FOLLOWING THE LAST BIOTIN ADMINISTRATION. |
| Transport Temperature: | Refrigerated |
| Stability (collection to initiation of testing): |
Ambient, 15 – 25 °C: Unacceptable Refrigerated, 2 – 8 °C: 24 Hours Frozen, (- 15) – (- 25) °C: 12 Months |
| Performed | Monday through Sunday |
| Analytic Time | 1 Day |
| CPL Code | 84484 |
Swab Sample Collection for 68_8800 CT_NG Quick Reference
cobas® CT/NG Swab Sample Collection for the cobas® 6800/8800 Systems
cobas® PCR Media Uni Swab Sample Kit
P/N: 07958030190
|
Anorectal |
Endocervical |
Oropharyngeal |
Vaginal |
|
|
Woven Swab |
✓ | ✗ | ✓ | ✓ |
cobas® PCR Media Dual Swab Sample Kit
P/N: 07958021190
|
Anorectal |
Endocervical |
Oropharyngeal |
Vaginal |
|
|
Woven Swab |
✓ | ✗* | ✓ | ✓ |
|
Flocked Swab |
✗ | ✓ | ✗ | ✗ |
*The woven swab is used to remove excess mucus from the cervical os and surrounding mucosa prior to endocervical specimen collection with the flocked swab.
|
Woven Swab
|
Flocked Swab
|
COBAS is a trademark of Roche. © 2021 Roche. MC-US-10189-1021
Roche Diagnostics Corporation
9115 Hague Road
Indianapolis, Indiana 46256
diagnostics.roche.com
Roche Transition: STI Testing
Effective September 6, 2023, East Side Clinical Laboratory will be transitioning Chlamydia trachomatis (CT), Neisseria gonorrhoeae (NG) and Trichomonas vaginalis (TV) testing to the Roche COBAS 6800 platform. In addition, we will also be offering testing for Mycoplasma genitalium (MG).
This transition phase will include the following changes to methodologies and collection devices:
| Current | New | |
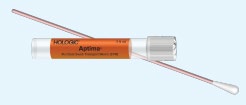
| Testing Platform | Hologic Aptima® | Roche COBAS® 6800/8800 |
| Methology | Transcription-Mediated Amplification and Hybrid Capture | Real-time Polymerase Chain Reaction (PCR) |
| Collection Device |
Aptima® Urine (Yellow Aptima)
|
COBAS® PCR Urine Kit collection devices COBAS® PCR Media Dual Swab Kit collection devices
|
- Both methods are FDA-approved nucleic-acid amplification technologies (NAAT) a. The Roche method is qualitative real-time polymerase chain reaction (PCR). Whereas, Hologic uses qualitative target capture and Transcription-Mediated Amplification (TMA), an alternated NAAT method.
- The Roche method is considered analytically equivalent to other NAAT methods according to CDC and ESCL evaluations.
- The Roche method has been confirmed to be highly sensitive and specific.
- The collection instructions and handling of the Roche collection swab & urine are similar to prior method collection, and the stability is equivalent.
Please contact your East Side Clinical Laboratory Sales Representative with any questions.
New testing will now require the COBAS PCR media collection devices
The transition will include replacing current collection device of Aptima® Urine (Yellow Aptima), Aptima® Multitest (Orange Aptima) and Aptima® Unisex (Blue Aptima) to COBAS® PCR Urine Kit and COBAS® PCR media Dual Swab Kit.
Clients will no longer be supplied with Aptima® Urine (Yellow Aptima), Aptima® Multitest (Orange Aptima) and Aptima® Unisex (Blue Aptima) collection devices after current inventory is depleted.
New orders will be fulfilled with COBAS® PCR Urine Kits and COBAS® PCR media Dual Swab Kit.
Distribution
Please contact your local ESCL facility for supplies. There will be no change in the ordering or distribution process.
NOTE: During the transition, ESCL will continue to perform testing on Aptima® Urine (Yellow Aptima), Aptima® Multitest (Orange Aptima) and Aptima® Unisex (Blue Aptima) collection devices until fully moved over to the Roche COBAS PCR Urine and Dual Swan Kit collection devices.
ESCL assures clients this transition causes no disruption of service. We believe this transition will improve testing for our clients, reduce instrumentation redundancy, as well as provide the opportunity for clients to test for Mycoplasma genitalium.
Order codes and test names will remain the same for testing. Mycoplasma genitalium has been added to the order code list.
| Collection Type | Acceptable Specimen Type | Order Code | Name |
| Swab | Vaginal, Endocervical, (ThinPrep) Cervical |
4179 | CHLAMYDIA TRACHOMATIS, NAAT |
| 4180 | NEISSERIA GONORRHOEAE, NAAT | ||
| 5650 | TRICHOMONAS VAGINALIS, NAAT | ||
| 4181 | C. TRACHOMATIS & N. GONORRHOEAE, NAAT | ||
| 5780 | C. TRACHOMATIS & N. GONORRHOEAE & TRICHOMONAS VAGINALIS, NAAT | ||
|
Vaginal, Endocervical, Throat |
53651 | MYCOPLASMA GENITALIUM, NAAT | |
| 5740 | C. TRACHOMATIS & N. GONORRHOEAE, NAAT (THROAT) | ||
| 5698 | C. TRACHOMATIS & N. GONORRHOEAE, NAAT (ANAL) | ||
| Urine | Urine | 4188 | CHLAMYDIA TRACHOMATIS, NAAT (URINE) |
| 4189 | NEISSERIA GONORRHOEAE, NAAT (URINE) | ||
| 5651 | TRICHOMONAS VAGINALIS, NAAT (URINE) | ||
| 53649 | MYCOPLASMA GENITALIUM, NAAT (URINE) | ||
| 4187 | C. TRACHOMATIS & N. GONORRHOEAE, NAAT (URINE) | ||
| 5649 | C. TRACHOMATIS & N. GONORRHOEAE & TRICHOMONAS VAGINALIS, NAAT (URINE) |
FDA Approved Status
The performance characteristics for the Roche COBAS® CT/NG and TV test have been evaluated and approved by the FDA for COBAS® PCR Urine Kit in male and female patients. ESCL has verified these performance characteristics.
Collection instructions for devices can be located below:
Please contact your East Side Clinical Laboratory Sales Representative with any questions.
cobas®️ STI Testing Sample Collection with the cobas®️ PCR Media Kits
URINE SAMPLE COLLECTION
Prior to sampling, the patient should not have urinated for at least one hour. Given that collection of larger volumes of urine may reduce test sensitivity, please direct the patient to provide first-catch urine (approximately 10 to 50 mL of the initial urine stream) into a urine collection cup (not provided).
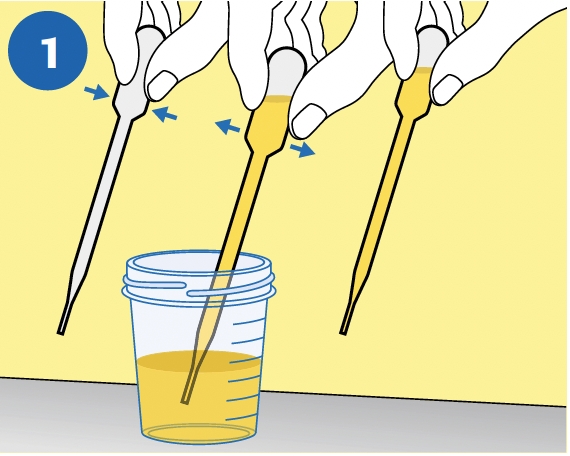 |
PIPETTE: Immediately transfer the urine into the cobas® PCR Media Tube using the provided disposable pipette. NOTE: If the urine specimen cannot be transferred immediately, it can be stored at 2ºC to 30ºC for up to 24 hours. |
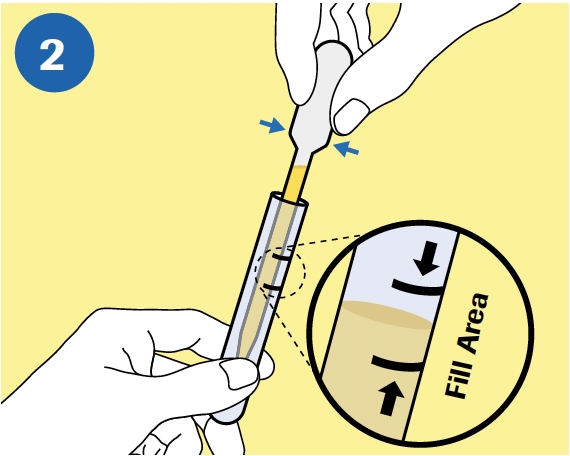 |
TRANSFER: The correct volume of urine has been added when the fluid level is between the two black lines on the tube label. |
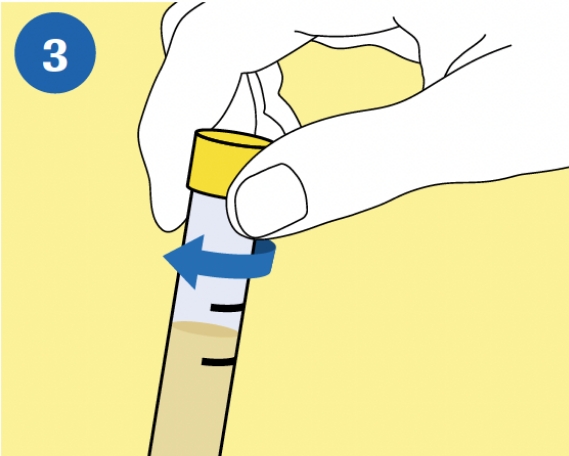 |
CAP: Tightly re-cap the cobas® PCR Media Tube. |
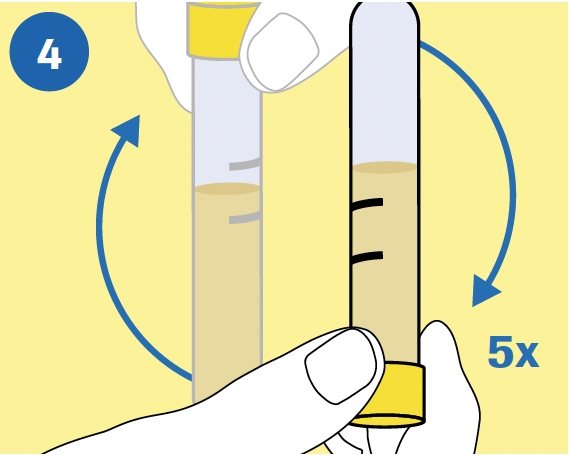 |
MIX: Invert the tube 5 times to mix. The specimen is now ready for transport. |
VAGINAL SWAB SAMPLE COLLECTION
NOTE: DO NOT pre-wet the swab in cobas® PCR Media before collection.
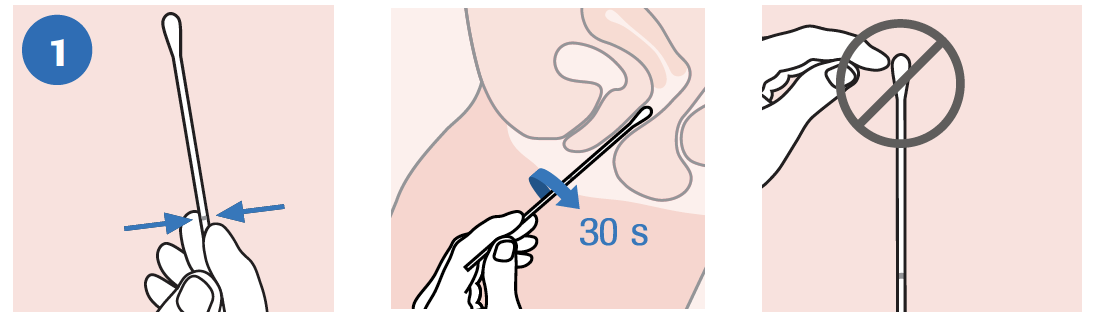 |
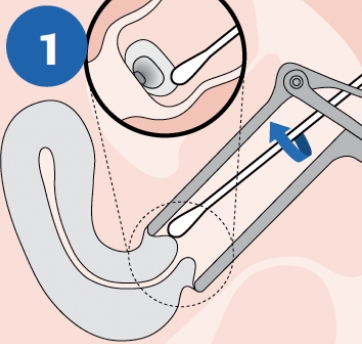
| COLLECT: To collect the specimen, hold the woven swab with the scoreline above your hand and insert the swab about 5 cm (2 inches) into the vaginal opening. Gently turn the swab for about 30 seconds while rubbing the swab against the walls of the vagina. Withdraw the swab carefully. Do not let the swab touch any surface before placing it into the collection tube. |
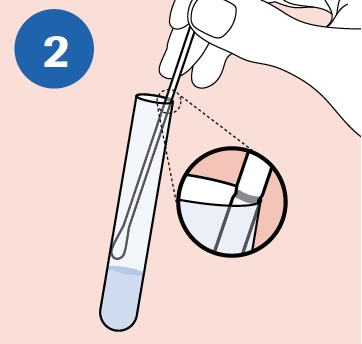 |
ALIGN: Remove the cap from the cobas® PCR Media Tube and lower the swab specimen into the tube until the visible scoreline on the swab is aligned with the tube rim. |
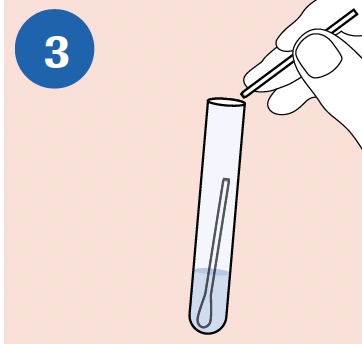 |
BREAK: Carefully leverage the swab against the tube rim to break the swab shaft at the scoreline. |
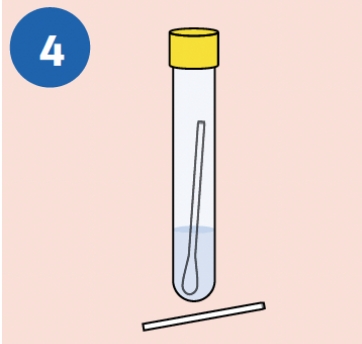 |
CLOSE: Tightly re-cap the cobas® PCR Media Tube. The specimen is now ready for transport. Discard the top portion of the swab |
VAGINAL SWAB SAMPLE COLLECTION
NOTE: DO NOT pre-wet the swab in cobas® PCR Media before collection.
 |
CLEAN: Using the woven swab, remove excess mucus from the cervical os and surrounding mucosa. Discard swab after cleaning. NOTE: Cleaning excess mucus from the cervical os is required to ensure an adequate sample is obtained for processing. |
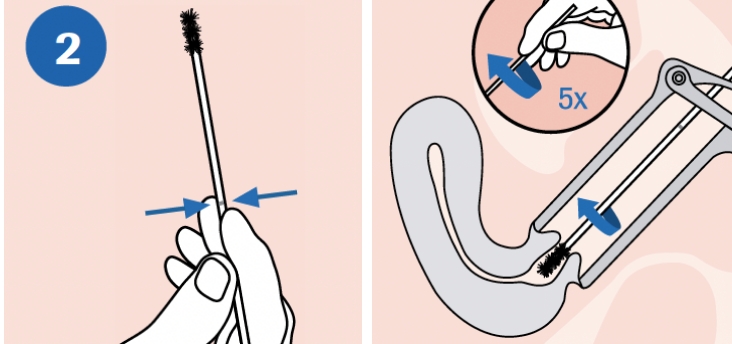 |
COLLECT: To collect the specimen, hold flocked swab with the scoreline above your hand and insert into the endocervical canal. Gently rotate the swab 5 times in one direction in the endocervical canal. Do not over-rotate. Carefully withdraw the swab, avoiding any contact with the vaginal mucosa. |
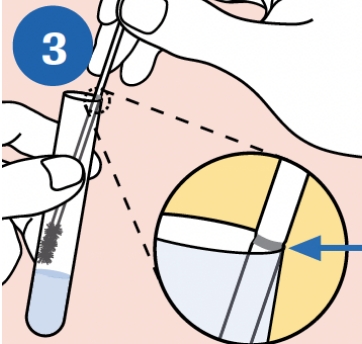 |
ALIGN: Remove the cap from the cobas® PCR Media Tube and lower the swab specimen into the tube until the visible scoreline on the swab shaft is aligned with the tube rim. The bud of the swab should not be submerged into liquid prior to breaking the shaft. |
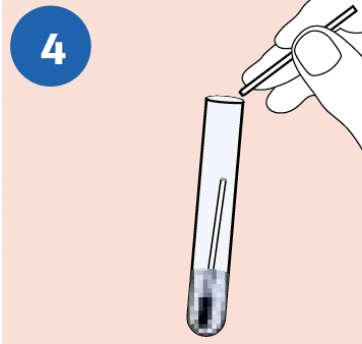 |
BREAK: Carefully leverage the swab against the tube rim to break the swab shaft at the scoreline. |
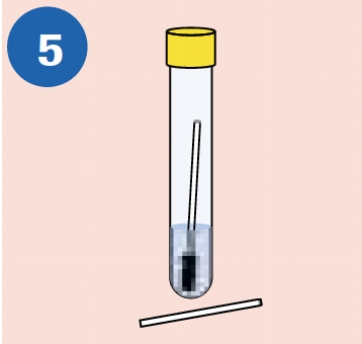 |
CLOSE: Tightly re-cap the cobas® PCR Media Tube. The specimen is now ready for transport. Discard the top portion of the swab. |
|
Specimen |
Collection and Transport Kit |
Sample Stability |
Testing Volume |
|
Male & Female Urine |
cobas® PCR Urine Sample Kit |
12 months |
850 μL |
|
Endocervical |
cobas® PCR Media Dual Swab Sample Kit |
12 months |
400 μL |
|
Vaginal |
cobas® PCR Media Uni Swab Sample Kit cobas® PCR Media Dual Swab Sample Kit |
12 months |
400 μL |
URINE SAMPLE COLLECTION TIPS
- Prior to sampling, the patient should not have urinated for at least one hour.
- This sample is a first-catch sample, not a mid-stream collection such as is used for urine culture.
- Not a lot of urine is needed; collect 10 - 50 mL of urine.
URINE SPECIMEN TRANSPORT AND STORAGE
- Ensure that the cap is tightened when closing the cobas® PCR Media Tube.
- Urine specimens must be transferred into the cobas® PCR Media Tube (stabilized) immediately. If specimens cannot be transferred immediately, they can be stored at 2ºC to 30ºC for up to 24 hours.
- Transport and store the cobas® PCR Media Tube containing the stabilized urine specimen at 2ºC to 30ºC. Stabilized urine specimens are stable at 2ºC to 30ºC for up to 12 months.
ENDOCERVICAL AND VAGINAL SWAB SPECIMEN COLLECTION TIPS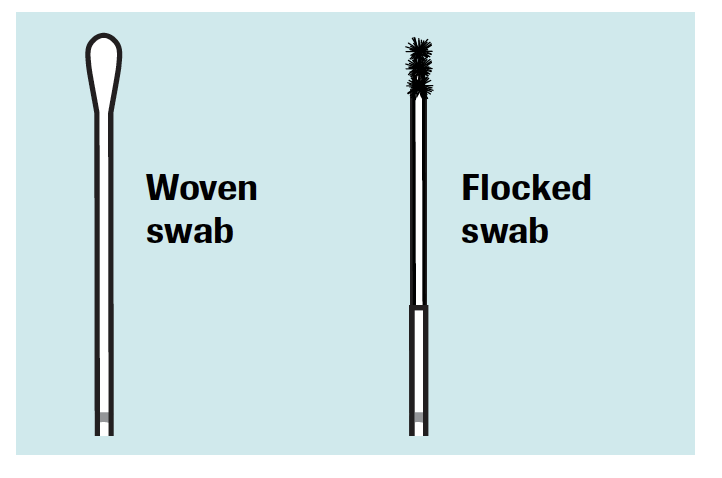
- Vaginal lubricants, speculum jellies, creams, and gels containing carbomer(s) may interfere
- with the test and should not be used during or prior to sample collection.
- If the collected specimen contains excess blood (specimen has a red or brown color), it should be discarded and not used for testing.
- Avoid contact of the cobas® PCR Media with the skin, eyes or mucous membranes. If contact does occur, immediately wash with large amounts of water.
- For endocervical sample collection with the cobas® PCR Media Dual Swab Kit, use the woven swab for cleaning and the flocked swab for sample collection.
- For vaginal sample collection with the cobas® PCR Media Uni or Dual Swab Kits, use only
the woven swab for sample collection. Discard the flocked swab.
SWAB SPECIMEN TRANSPORT AND STORAGE
- Ensure that the cap is tightened when closing the cobas® PCR Media Tube.
- Transport and store the cobas® PCR Media Tube containing the collection swab at 2°C to 30°C.
- The specimen should only contain one swab and may be rejected if the tube contains no swab or two swabs.
COBAS is a trademark of Roche.
All other product names and trademarks are the property of their respective owners.
© Roche 2020. MC-US-02272-0720
Roche Diagnostics Corporation
9115 Hague Road
Indianapolis, IN 46256
diagnostics.roche.com
Helicobacter pylori Urea Breath Test Manufacturer Change
The Gulf Coast ScientificTM PyloPlus® Urea Breath Test (UBT) for detection of Helicobacter pylori will replace the Meridian/OtsukaTM BreathTek®UBT. The Meridian/OtsukaTM assay has been discontinued by the manufacturer. Validation has determined that:
- The PyloPlus® UBT and BreathTek® UBT use the same methodology, infrared spectrophotometry of CO2 isotopes in breath samples before and after ingestion/metabolism of 75mg of 13C Urea in a citric acid solution.
- The performance characteristics of the PyloPlus®UBT and BreathTek®UBT tests are equivalent.
- As results are qualitative (Positive/Negative), the result reporting and reference interval are unchanged.
- There are minor changes to the collection kit and instructions. Those are given in detail below.
Effective March 15, 2023, distribution will begin transitioning to PyloPlus®UBT collection kits. Clients may be supplied with the PyloPlus® or BreathTek®UBT collection kits depending on availability of supplies. During this transition, the BreathTek®UBT collection devices will be run on the Meridian/Otsuka platform and incoming PyloPlus®UBT will be run on the Gulf Coast Scientific spectrophotometer platform without delays to testing.
| Name | Test Code |
| H Pylori Breath Test (BREATH) | 5173 |
| H Pylori Pediatric Stool (HPYLPED) | 18321 |
Pediatric Specimens
Gulf Coast Scientific PyloPlus® Urea Breath Test is an FDA-cleared test on adult patients 18 years or older. The Gulf Coast Urea Breath Test is not currently FDA approved for pediatric use. For patients <18 years of age, please perform the pediatric Helicobacte rpylori stool antigen test, test code 18321, by collecting a small sample of stool in a sterile cup.
Gulf Coast ScientificTM PyloriPlus® UBT Procedure
1. Materials provided in each PyloPlus Kit:

- One pouch of 13C-urea powder
- One packet of citric acid flavoring
- Test instructions
- One Quick Reference Instructions (QRI)
- Two breath collection bags, one blue bag for the BASELINE sample and one red bag for the POST sample
- One drinking straw
- One sample transport bag
2. Materials needed, but not provided
- A 15-minute timer
- Potable water
- Cup able to measure 100mL of water
3. Patient Preparation
- The patient should have fasted at least 1 hour before administering the PyloPlus Kit.
- Antimicrobials/antibiotics, Proton Pump Inhibitors (PPI), Bismuth preparations and Histamine Receptor antagonists (HRA) are also known to suppress H.pylori and may produce false-negative results. Any positive, while on those preparations should be considered a true positive. Upon the advice of their provider, patients should refrain from taking:
i. Antibiotics (e.g., Flagyl, Metronidazole): 2 weeks
ii. PPI’s (e.g., Nexium, Prevacid, Zegerid and Prilosec): 2 weeks
iii. Bismuth preparations (e.g., Pepto-Bismol, Kaopectate and Pink Bismuth): 2 weeks
iv. HRA’s (e.g., Pepcid, Zantac, Axid, Tagamet): 2 days
4. Step-By-Step Procedure
- Time intervals listed in the following step-by-step procedure are critical.
- Verify that the patient has been prepared for the test as specified above. If the patient is not appropriately prepared, contact the ordering clinician for further instructions.
- Open the PyloPlusKit, which should contain all the materials listed in 2.1. Label each breath collection bag to maintain patient identification using a felt tip permanent marker. Include the specimen date and time of collection.
- Collect the BASELINE breath sample according to the following steps:
i. Pick up the blue breath collection bag.
ii. Remove the twist-off cap from the mouth piece of the breath collection bag.
iii. Instruct the patient to: (1) breathe normally; (2) take a deep breath then hold their breath for 10 seconds; (3) partially exhale into the room, before fully exhaling into the mouthpiece of the bag.
iv. Replace the cap firmly on the mouthpiece of the bag.
v. NOTE: if the patient has not held their breath for 10 seconds, or does not fill the bag completely, there is a possibility a test result will not be obtained.
vi. NOTE: The bag is not fully closed if the cap does not click into place. Not fully closing the bag may cause the breath sample to leak, invalidating the result. - Prepare the 13C-urea solution no more than 50 minutes before administering it to the patient. Urea slowly decomposes in water.
i. Pick up the citric acid flavoring packet and tear open. Place contents of flavoring packet into 13C-urea pouch by tearing open the pouch and carefully pouring contents of the flavoring packet into the open 13C-urea pouch.
ii. Add approximately 100mL drinking water (about 1/3 full) to the 13C-urea pouch.
iii. Close the Ziplock feature of the 13C-urea pouch securely and shake the mixture for 30 seconds.
iv. Instruct the patient to drink all of the drug solution with the straw provided directly from the 13C-urea pouch, without stopping. Advise the patient NOT to ‘rinse’ the inside of their mouth with the drug solution before swallowing. Discard the straw after the patient has finished drinking the drug solution. - Set the timer for 15 minutes. The patient should sit quietly and should not eat, drink, or smoke during the 15-minute interval.
- After 15 minutes have elapsed, pick up the red (pink) breath collection bag. Collect the POST-SAMPLE breath sample according to the instructions above
References
PyloPlus® UBT Kit for PyloPlus® UBT System Breath Test for Detection of H. pylori. Package Insert. Gulf Coast Scientific TM. REV_B
Neisseria Gonorrhoeae Culture & Sensitivity
Effective April 10, 2023 East Side Clinical Laboratory will offer antimicrobial susceptibility testing on positive Neisseria Gonorrhoeae cultures.
Principle
Neisseria gonorrhoeae is a sexually transmitted organism usually recovered from genital (vaginal, cervical, urethral, prostate, semen, etc.) sources. It can, however, be isolated from various other mucosal body sites (eye, throat, anal, synovial fluid). All genital cultures automatically incorporate a screen for N. gonorrhoeae in the culture. If the clinician suspects an infection from N. gonorrhoeae in a non-genital site, a gonorrhoeae culture should be performed. A gonorrhoeae culture is also performed when a clinician specifically orders a culture exclusively for N. gonorrhoeae from a genital site.
Specimen Collection, Transport & Handling
Specimens should be collected utilizing an eSwab. Acceptable Sources:
1. Endocervical
No use of lubricant during collection. Wipe cervix clean of mucus. Rotate swab, obtain exudate from endocervical glands. If no exudate, insert into canal and rotate swab.
2. Vaginal (not optimal)
Insert swab into vagina. Collect discharge or secretions from mucosa in vaginal canal.
3. Urethral
- Collect 1-2 hrs or more after urination.
- Female: stimulate discharge by massaging urethra against pubic symphysis through vagina. Collect discharge with swab. If discharge cannot be collected, wash external urethra with Betadine soap and rinse with water. Insert a urogenital swab, rotate. Leave if 1-2 seconds, and withdraw.
- Male: insert thin urogenital swab 2-4 cm into endourethra, rotate. Leave for 1-2 seconds, and withdraw.
4. Epididymis
Aspirate material, and eject into sterile container.
5. Pharyngeal
Push down tongue gently with a depressor. Extended swab between tonsillar pillars and behind uvula. Avoid touch surfaces of mouth. Sweep back and forth across posterior pharynx.
6. Rectal (free of stool)
Pass tip of swab approximately 2 cm beyond anal sphincter. Rotate, and withdraw.
7. Eye
- Collect purulent material on a swab.
- All eye cultures from newborns [up to 1 month old] incorporate are to be screened for gonorrhoeae.
8. Synovial fluid
Ordering Information:
| Name | Test Code |
| Neisseria Gonorrhoeae Culture | 5006 |
Specimen Requirements: ESWAB
Transport Temperature: Room Temperature
Stability: 24 hours (For best results submission within less than 12 hours recommended)
Expected time to result: 3 days
Unsuitable Specimens: Frozen specimens
CPT Codes: 87081 (additional CPT codes will apply if microbial testing is needed)
A panel of antibiotics recommended by the Clinical Laboratory Standards Institute (CLSI) includes:
- Cefepime
- Cefixime
- Cefotaxime
- Ceftriaxone
- Ciprofloxacin
- Penicillin
- Tetracycline
***Please indicate source on collection device and on requisition***
Please contact your East Side Clinical Laboratory Sales Representative with any questions.
Discontinuation of Throat Swab for Flu
Effective April 30, 2023, East Side Clinical Laboratory will discontinue accepting throat swabs for Influenza A + B testing. Beginning May 1, 2023, testing for Influenza A + B will only be performed utilizing a nasal or nasopharyngeal collection. By discontinuing throat collection, we can utilize a nucleic acid amplification test (NAAT) which detects up to 42% more true positives when compared to a rapid antigen test (RADT), providing the best quality testing for patients.
Please contact your East Side Clinical Laboratory representative with any questions.
Acceptable Specimen & Swab Types for Respiratory Testing
Group A strep:
- Specimen types:
- Throat - Container types:
- Dry culturette swab (CANNOT be combined with a second test)
- E-swab (can be combined with other tests)
Flu:
- Specimen types:
- Nasopharyngeal (preferred)
- Nasal - Container types:
- Dry culturette swab (CANNOT be combined with a second test)
- E-swab (can be combined with other tests)
- VTM (can be combined with other tests)
COVID:
- Specimen types:
- Nasopharyngeal (preferred)
- Nasal
- Oropharyngeal - Container types:
- VTM (preferred)
- E-swab
MULTIPLEX (FLU, RSV & SARS-CoV-2):
- Specimen types:
- Nasopharyngeal (preferred)
- Nasal - Container types:
- VTM
COVID SWABS ARE DISCARDED ONCE RESULTS ARE FINALIZED.
Other respiratory testing CANNOT be added on later once PCR testing has occurred
2022
Monkeypox Virus (MPX) by PCR
Effective July 25, 2022, East Side Clinical Laboratory is pleased to announce the availability of Monkeypox Virus by PCR testing.
Monkeypox Virus PCR test methodology is real-time PCR utilizing the CDC Non-variola Orthopoxvirus Real-Time PCR Primer and Probe Set. The assay detects non-variola Orthopoxvirus DNA in clinical specimens. Although this assay does not differentiate monkeypox virus from other Orthopoxvirus including vaccinia, cowpox, camelpox, ectromelia or gerbilpox virus a positive result with this assay in the United States is most likely due to monkeypox virus; however, potential exposure to other Orthopoxviruses should be considered.
The test results are intended to be utilized in conjunction with clinical presentation, epidemiological data, other diagnostic test results, and vaccination and exposure history. As monkeypox virus infections can present with rashes/lesions similar to that of varicella zoster virus, herpes simplex virus, and other sexually transmitted infections, consider testing for other pathogens as clinically indicated.
Test Code: 5900
Ordering recommendation: For detection of non-variola Orthopoxvirus, including monkeypox virus, DNA in clinical specimens.
Specimen Requirements: Double-Dry swab (polyester, rayon, or dacron) collection from skin lesion. Two swabs should be submitted to ensure adequate material is sampled. Break off or cut the tip of each swab into a sterile tube or container. Each swab should be placed in a separate tube or container. Freezing sample is strongly recommended.
Unsuitable specimen: Single swab. Cotton swab. Swabs with wooden or wire shafts. Swabs in media designed for preservation and/or transport.
Transport temperature: Frozen
Stability: 7 days refrigerated; 1 month frozen.
CPT codes: 87593
For questions, contact your East Side Clinical Laboratory Customer Service Representative.
Laboratory Testing Guidelines
- Swab or brush the pustule/lesion vigorously with two separate sterile swabs.
- Collect at least two swabs from the same lesion.
- If possible, sample paired specimens from multiple lesions on different parts of the body and with differing appearances. CDC suggests 2-3 lesions total.
- Use a sterile nylon, polyester, or dacron swab with a plastic or thin aluminum shaft.
- Do not use other types of swabs. Do NOT use cotton swabs.
- Break off or cut the tip of each swab into a sterile tube or container.
- Place each swab into a separate dry sterile tube or container and ensure that the tube/container is tightly sealed and leak proof.
- Do NOT add transport media to specimen.
- Multiple specimens collected on a single patient should be submitted separately.
- Each individual specimen submitted for Monkeypox virus testing should be accompanied by its own separate requisition and transported in its own sealed bag.
-
Refrigerate (2–8°C) or freeze (-20°C or lower) specimens within an hour after collection.
-
Freezing is strongly recommended.
-
Ship frozen (preferred) or refrigerated (acceptable). To maintain optimum specimen viability, transport the specimen to the laboratory as soon as possible.
Monkeypox: Interim Guidance from the CDC for Healthcare Providers
Clinical features
- Caused by Monkeypox virus (MPXV)
- Two clades: Central African (CAC) & West African (WAC). WAC is less virulent and is the predominant clade circulating in the US.
- Orthopox genus, which includes variola/smallpox, cowpox. MPXV is NOT related to varicella/chickenpox/shingles.
- Demonstrates extraordinary resistance to drying, heat, and pH, which leads to environmental persistence. Materials with MPX virus may remain infectious for months to years.
- Incubation period - 6 -13 days, up to 21 days.
- Signs/Symptoms
- Fever, headache, swollen lymph nodes (which may differentiate it from smallpox), respiratory symptoms, GI symptoms, including diarrhea.
- Characteristic Rash - See photos and complete description, below
- May look like pimple or blister
- Appears on face, inside of mouth, genitals, and other body parts
- Labs – Leukocytosis, elevated AST & ALT, low BUN, low Albumin
- Differential diagnosis - Smallpox, chickenpox/shingles, measles, bacterial skin infections, scabies, medication allergies, HSV, and syphilis.
-
Spread
-
Direct contact with the rash, respiratory secretions during prolonged face to face contact, intimate contact, fomites, placental transfer, animals handling.
-
Possible for up to several weeks.
-
-
Prevention
-
Avoid contact, good hygiene, PPE
-
Vaccine – JYNNEOS/Imvanex: inactive virus, FDA approved
-
-
Complications – Bacterial infection, sepsis, dehydration, diarrhea, encephalitis, death.
-
Treatment – None, but smallpox anti-viral (TPOXX/tecovirimat) may be indicated for vulnerable populations.
Criteria to Guide Evaluation of Monkeypox Cases
| Suspect Case | New characteristic rash (See complete description, below). |
OR | Meets one of the epidemiological criteria and has a high clinical suspicion for Monkeypox. (See below). |
| Probable Case | No suspicion of other recent Orthopoxvirus exposure (e.g., Vaccinia virus in ACAM2000 vaccination). | OR | Demonstration of the presence of orthopoxvirus DNA. |
| Confirmed Case | Demonstration of the presence of Monkeypox virus DNA by polymerase chain reaction testing or Next-Generation sequencing of a clinical specimen. | OR | Demonstration of Monkeypox virus in culture from a clinical specimen. |
Epidemiologic Criteria – Within 21 days of illness onset:
- Reports having contact with a person or people with a similar appearing rash or who received a diagnosis of confirmed or probable monkeypox OR
- Had close or intimate in-person contact with individuals in a social network experiencing monkeypox activity, this includes men who have sex with men (MSM) who meet partners through an online website, digital application (“app”), or social event (e.g., a bar or party) OR
- Traveled outside the US to a country with confirmed cases of monkeypox or where Monkeypox virus is endemic OR
- Had contact with a dead or live wild animal or exotic pet that is an African endemic species or used a product derived from such animals (e.g., game meat, creams, lotions, powders, etc.)
Exclusion Criteria – A case may be excluded as a suspect, probable, or confirmed case if:
- An alternative diagnosis* can fully explain the illness OR
- An individual with symptoms consistent with monkeypox does not develop a rash within 5 days of illness onset OR
- A case where high -quality specimens do not demonstrate the presence of Orthopoxvirus or Monkeypox virus or antibodies to orthopoxvirus.
Characteristic Rash
- Deep-seated and well-circumscribed lesions, often with central umbilication.
- Lesion progression through specific sequential stages—macules, papules, vesicles, pustules, and scabs.
- May sometimes be confused with other diseases that are more commonly encountered in clinical practice (e.g., secondary syphilis, herpes, and varicella zoster).
- Historically, sporadic accounts of patients co-infected with Monkeypox virus and other infectious agents (e.g., varicella zoster, syphilis) have been reported, so patients with a characteristic rash should be considered for testing, even if other tests are positive.
The outbreak caused by MPXV is rapidly evolving, and we will continue to monitor the situation and update you as new information becomes available.
Extensive additional information on monkeypox can be found at the CDC’s website.
References:
Collection, Storage, and Shipment of Specimens for Monkeypox Diagnosis. CDC.
https://www.cdc.gov/poxvirus/monkeypox/clinicians/prep-collection-specimens.html
Case Definitions for Use in the 2022 Monkeypox Response. Centers for Disease Control and Prevention website. Accessed June 2022. https://www.cdc.gov/poxvirus/monkeypox/clinicians/case-definition.html
U.S. Monkeypox Outbreak 2022: Situation Summary. Centers for Disease Control and Prevention website. Accessed June 2022.
US Monkeypox outbreak 2022. CDC’s 2022 U.S. Map & Case Count.
https://www.cdc.gov/poxvirus/monkeypox/response/2022/us-map.html
Brown K, Leggat PA. Human Monkeypox: Current State of Knowledge and Implications for the Future. Trop Med Infect Dis. 2016; 1(1):8. Published 2016 Dec 20. doi:10.3390/tropicalmed1010008
KD Reed, et al., The detection of monkeypox in humans in the Western Hemisphere. N Engl J Med. 2004. 350, 342–350. https://pubmed.ncbi.nlm.nih.gov/14736926/
SARS-CoV-2 Omicron Variant of Concern
On November 30, 2021, the U.S. SARS-CoV-2 Interagency Group (SIG), which includes the Centers for Disease Control and Prevention (CDC), the National Institutes of Health, the Food and Drug Administration (FDA), and Health and Human Services (HHS), classified the Omicron variant as a Variant of Concern. The current understanding of the Omicron VOC at this time is as follows:
- Preliminary data from South Africa suggest that the mutations to the receptor binding protein of the variant virus will confer increased infectivity.
- Preliminary studies indicate that there are no unusual symptoms associated with Omicron variant infection, and as with other variants, some patients are asymptomatic. Symptoms may be milder in persons who have been vaccinated or previously infected with SARS CoV-2.
- The presence of mutations in the SARS-CoV-2 virus in a patient sample has the potential to impact test performance. The impact of mutations on a test's performance is influenced by several factors, including the sequence of the variant, prevalence of the variant in the population, and the design of the test including the analytic target. Indeed, tests which interrogate for a single target are particularly vulnerable.
Laboratory evaluation of SARS-CoV-2, including the Omicron Variant of Concern, at East Side Clinical Laboratory:
- All high-throughput SARS-CoV-2 molecular assays performed at East Side Clinical Laboratory are designed to interrogate for multiple targets of the viral genome and have received emergency-use authorization by the FDA.
- Based on in silico analysis, all molecular assays are expected to detect the Omicron variant of concern.
- For some assays, including the Thermo Fisher Taq Path PCR assay, a specific deletion in the spike (S) gene (Δ69-70) in the Omicron variant results in an S-gene dropout, also referred to as an S-gene target failure (SGTF).
- The S-gene target failure pattern may be identified in the Omicron variant (BA.1 sub-lineage), but this pattern is also seen in previously identified variants (e.g., Alpha).
- Since these tests are designed to detect multiple genetic targets, the overall test sensitivity is not significantly impacted.
- The S-gene target failure pattern provides a signal that the Omicron variant (BA.1) may be present and that the isolate may be suitable for sequencing and/ or other public health considerations.
- The S-gene target failure pattern does not necessarily mean that an individual with SARS-CoV-2 has the Omicron variant.
- For clients who have concerns about antigen or other point of care assays that they provide, the assay manufacturer can be contacted for information about expected analytical performance impact.
References:
- New SARS-CoV-2 Variant of Concern Identified: Omicron (B.1.1.529) Variant. Health Alert Network 00459. December 1, 2021.
- SARS-CoV-2 Viral Mutations: Impact on COVID-19 Tests. U.S. Food and Drug Administration. December 6, 2021.
- Technical Brief: Predicted Impact of Variants on Abbott SARS-CoV-2/COVID-19 Diagnostic Tests. Abbott. November 26, 2021
- Impact of SARS-CoV-2 mutations (including Omicron) on the cobas® SARS-CoV-2 and cobas® SARS-CoV-2 & Influenza A/B Tests for use on the cobas® 6800/8800 Systems and the cobas® SARS-CoV-2 and cobas® SARS-CoV-2 & Influenza A/B Tests for use on the cobas® Liat® System. Roche Client Bulletin. November 30, 2021.
- Thermo Fisher Scientific Confirms Detection of SARS-CoV-2 in Samples Containing the Omicron Variant with its TaqPath COVID-19 Tests. Thermo Fisher Client Bulletin. November 29, 2021.
NEW SARS-CoV-2, Influenza A+B, RSV Panels
Changes in Estimated Glomerular Filtration Rate (eGFR) Calculation: Removal of Race-Based Adjustment
RESPIRATORY SYNCYTIAL VIRUS (RSV)/HUMAN METAPNEUMOVIRUS (hMPV)
Effective October 13, ESCL will begin performing RSV/hMPV by NAAT.
The Solana RSV + hMPV Assay is a qualitative in vitro diagnostic test for the detection and differentiation of RSV and hMPV viral RNA in nasal and nasopharyngeal swabs from patients with signs and symptoms of respiratory infection. This test is intended for use as an aid in the differential diagnosis of RSV and hMPV viral infections in humans in conjunction with clinical and epidemiological risk factors. This test is not intended to differentiate the two subtypes of RSV or the four genetic sub‐lineages of hMPV.
RSV: Human respiratory syncytial virus (RSV) is a negative single‐stranded RNA virus of the family Paramyxoviridae. RSV is the major cause of lower respiratory tract infection and hospital visits during infancy and childhood.
hMPV: Human metapneumovirus (hMPV) is a negative single‐stranded RNA virus of the family Paramyxoviridae first isolated in 2001 in the Netherlands. hMPV may be the second most common cause (after RSV) of lower respiratory infection in young children. Compared with RSV, infection with hMPV tends to occur in slightly older children and produce disease that is less severe. Co‐infection with both viruses can occur and is generally associated with more severe disease.
Test Code: 11720
Specimen Requirements: VIRUS TRANSPORT MEDIA OR EQUIVALENT.
ESWABs and Dacron or Rayon swabs are also acceptable.
Transport temperature: Refrigerated
Stability: VTM: 6 days refrigerated; eSwab 48 hours refrigerated
Result Interpretation: Negative
CPT code: 87634; 87798
For questions contact your East Side Clinical Laboratory representative
Sonic Healthcare USA is announced as a Laboratory Testing Partner for National K-12 School Testing Program
Austin, TX – April 19, 2021
Sonic Healthcare USA (Sonic), a leading provider of laboratory services in the United States, was recently included as a laboratory testing partner for the Rockefeller Foundation’s K-12 National Testing Action Program (NTAP). The NTAP program introduces school districts in the United States to leading testing companies supporting COVID-19 testing for K-12 students.
Over the last year, Sonic has been one of the leading commercial laboratories performing COVID-19 testing throughout its network of clinical laboratories and pathology practices in the United States. Sonic continues to focus on and support specific underserved patient populations and government-funded programs in partnership with local government and public health agencies to improve testing access for these communities.
Sonic is expanding testing capacity for K-12 testing programs at the following laboratory locations: American Esoteric Laboratories, located in Memphis, TN; Bernhardt Laboratories, located in Jacksonville, FL; Clinical Labs of Hawaii, located in Honolulu, HI; Clinical Pathology Laboratories, located in Austin, TX; East Side Clinical Laboratory, located in Providence, RI; GPA Laboratories, located in Greensboro, NC; LMC Pathology Services, located in Las Vegas, NV; Seacoast Pathology, located in Exeter, NH; Sunrise Medical Laboratories, located in Hicksville, NY; and WestPac Labs, located in Santa Fe Springs, CA.
“We are honored and privileged to be included amongst the leading companies supporting COVID-19 testing efforts,” said Jerry Hussong, MD, MBA, Chief Executive Officer, Sonic Healthcare USA. “Supporting the reopening of schools is paramount to COVID-19 recovery efforts; it is our social responsibility to collaborate with public health officials, school officials, and other leading private and public sector organizations. Collectively, we can quickly respond to the needs of our local communities,” added Dr. Hussong.
About Sonic Healthcare USA
Sonic Healthcare USA is a subsidiary of Sonic Healthcare Limited, one of the world's largest medical diagnostic companies, providing laboratory services to medical practitioners, hospitals and community health services, with operations in eight countries, on three continents and providing care to over 100 million patients each year. Sonic Healthcare USA is a leading provider of state-of-the-art laboratory services and pathology practices throughout the USA with eight operating divisions and nearly eight thousand US- based employees, 330 Pathologists and serving over 30 million patients per year. Sonic Healthcare USA operates under a federated business model that emphasizes medical leadership and community-based testing services to provide outstanding quality and service to the doctors and patients that they serve. For more information, visit the Sonic Healthcare website at www.sonichealthcareusa.com.
MEDIA CONTACTS:
Sonic Healthcare USA
Dr. Jerry Hussong, MD, MBA
Chief Executive Officer
jhussong@sonichealthcareusa.com
512.531.2216
SARS-CoV-2 S Antibody (Spike Semi-Quantitative)
Effective February 8, 2021, ESCL is announcing availability of a Spike Antibody test for COVID-19. This immunoassay detects high affinity antibodies against spike (S) protein receptor binding domain (RBD). The assay is intended as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection.
This assay also identifies the immune response induced by vaccines targeting the SARS-CoV-2 Spike (S) protein receptor binding domain (RBD).
Test Code: 11359
Traceability: Internal Roche standard for anti-SARS-CoV-2 S consisting of monoclonal antibodies.
Specimen Requirements: Serum from an SST tube
Transport temperature: Refrigerated
Stability: 7 days refrigerated
Result Interpretation: <0.8 U/ml = negative, >=0.8 U/ml = positive
Positive Percent Agreement (PPA): 96.6% in samples >14 days post-PCR positive.
Negative Percent Agreement (NPA): 99.98% in potentially cross-reactive samples.
CPT code: 86769
For questions contact your East Side Clinical Laboratory representative
10 Risho Ave East Providence RI 02914
www.esclab.com
Issue Date: February 5, 2021
SARS-CoV-2 (COVID-19) Requisition
Announcement: No Swab Collections during COVID-19
Dear Providers,
East Side Clinical Laboratory is taking further steps to prevent the spread of COVID-19. Minimizing the risk of transmission among staff and between staff and patients as well as protecting people who are at higher risk for adverse health complications is of utmost concern to us.
In light of these concerns, we have implemented the following change to our practice to mitigate the impact of COVID -19:
We will not be providing nasal/throat swab collections (e.g. for influenza, group A strep, throat culture testing) at our Patient Service Centers or Independent Physician Offices during the ongoing COVID19 pandemic.
Note: Patients visiting a patient service center must wear a mask or face covering and must not be experiencing COVID -19 symptoms. Patients may not take their mask or face covering off while in our Patient Service Centers. We do not offer swab collection for COVID-19 testing.
Our mission is to provide the highest quality laboratory testing to our patients and clinical providers to aid in the diagnosis and treatment of disease.
For the safety of our patients and our staff, we cannot perform any specimen collections that would require patients to remove their masks or face coverings.
Thank you,
Walther M. Pfeifer, MD, FCAP Laboratory Director East Side Clinical Laboratory
Sonic Healthcare USA to Expand COVID-19 Testing Capacity in the United States in Collaboration with the NIH RADx Initiative
Austin, TX – September 2, 2020
Today, Sonic Healthcare USA (Sonic) was awarded a contract from the National Institutes of Health (NIH) Rapid Acceleration of Diagnostics Advanced Technology Platforms (RADx-ATP) initiative. The contract involves funding to expeditiously ramp up COVID-19 testing to 166,000 tests per day at nine of Sonic’s network of high-throughput laboratory testing locations, giving Sonic the capacity to service geographies across the United States.
Sonic was also selected by the U.S. Department of Health and Human Services (HHS) as one of two commercial laboratories to receive critical laboratory equipment to support the expansion of COVID-19 testing. Through these investments, aggressive expansion has begun at the following Sonic COVID-19 testing locations: American Esoteric Laboratories (AEL), located in Memphis, TN, Clinical Pathology Laboratories (CPL), located in Austin, TX and WestPac Labs located in Santa Fe Springs, CA. The added capacities will be coming online beginning this week.
While the NIH contract amplifies Sonic’s existing COVID-19 testing effort for all populations, a significant component includes improving testing access for underserved communities. Sonic has been supporting these populations directly and in partnership with local government and public health agencies. Funding from the RADx initiative will be used to develop comprehensive access with measurable coverage assessment for these populations. The RADx initiative complements Sonic’s current commitment to prioritize services to the most vulnerable and high-risk patients. Servicing these populations will continue to be a central part of our testing strategy.
“In response to the urgent pandemic demands, the partnership with the NIH and HHS, provides us access to technologies, instrumentation and resources necessary to rapidly increase the scope and expedite the timeline of our current capacity expansion plans,” said Dr. Jerry Hussong, MD, MBA, the Chief Executive Officer of Sonic HealthcareUSA. “The new testing capacity will increase patient access to COVID-19 diagnostic testing across the country with timely result delivery,” added Dr. Hussong.
This project is supported by the NIH Rapid Acceleration of Diagnostics (RADx) program and has been funded in whole or in part with federal funds from the National Institutes of Health Office of the Director, Department of Health and Human Services, under Contract No. 75N92020C00027.
Lyme Modified Two-Tier Test
Lyme Modified Two-Tier Test (MTTT)
Effective Monday, August 17, 2020, East Side Clinical Laboratory will offer testing for Lyme disease with the Modified Two-Tier Testing (MTTT) methodology. It is intended to be used in symptomatic patients suspected of having Lyme disease.
MTTT is a FDA cleared sensitive and specific alternative to Standard Two-Tier Testing (STTT) for Lyme disease. Unlike the STTT, the MTTT utilizes ELISA in both the first and second tiers, and does not utilize immunoblotting (Western Blot or Line Blot methods), detecting more acute Lyme disease cases compared with the traditional testing methods.
The first-tier is an ELISA which screens for B.burgdorferi VlsE/pepC10 IgG/IgM antibodies, with positives reflexing to the second tier.
The second-tier is an ELISA utilizing B.burgdorferi WCS (Whole Cell Sonicate antigens), comprised of specific assays for IgG and IgM, in which all results are individually reported with an Index Value, followed by comments and interpretations.
References:
- Updated CDC recommendation for Serologic Diagnosis of Lyme Disease can be found in MMWR/August 16,2019/Vol. 68/No.32 703 or at
- https:www.cdc.gov/mmwr/volumes/68/wr/mm6832a4.htm?s_cid=6832a4_w
Test Code: 25851
Specimen Requirements: Serum from SST or plain red top tube
Transport Temperature: Refrigerated
Stability: 10 days refrigerated
CPT codes: 88618-If reflexed, add 86617(x2)
Please contact your ESCL account representative with any questions.
Discontinuation of Serum and Urine Ketone Testing by Acetest and Replacement by Beta-Hydroxybutyrate
Effective July 1, 2020, East Side Clinical Laboratory has discontinued performing Acetest testing for serum and urine ketones due to discontinuation of reagent by the manufacturer and has been replaced with quantitative serum B-Hydroxybutyrate (6942).
The presence and degree of ketosis can be determined by measuring levels of Beta-hydroxybutyrate, which is the ketoacid present in the greatest amount in serum, accounting for 75% of the ketone bodies. During periods of ketosis, the relative percentage of Beta-hydroxybutyrate increases even more over the other two ketoacids (acetoacetate and acetone) and has been shown to be a better index of ketoacidosis including the detection of sub-clinical ketosis.
βHB levels have also been shown to be useful for assessing patients that may require additional insulin therapy following IV insulin drips for DKA. βHB may be helpful for establishing endpoints for IV therapy.
Discontinued tests with order codes:
|
6007 ACETONE (SERUM)
|
| 6035 ACETONE (URINE) |
Replacement test with order codes:
| 6942 BETA HYDROXYBUTURATE |
| Reference Range | 0.2-2.8 mg/dL |
| Specimen Type | Serum from SST , or Heparinized Plasma |
| Stability | When separated from cells; refrigerated 1 week; frozen 1 month |
| Storage | Refrigerated |
| Turnaround time | 1 day |
If you have any questions, please call East Side Clinical Laboratory at 800-455-8440
Or contact your East Side Clinical Laboratory representative.
Thank you for allowing us to continue to provide the highest quality laboratory testing services to you and your patients.
Reference Range Update: Complete Blood Count (CBC)
Effective April 30, 2020, East Side Clinical Laboratory (ESCL) will update reference ranges for the Complete Blood Count (CBC).
The new adult reference ranges reflect a Sonic Healthcare USA consensus which align with those of other large reference laboratories and hospital systems. The pediatric reference ranges align with those published by the American Association for Clinical Chemistry [Pediatric Reference Intervals, 7th ed. (2011)] and American Society for Clinical Pathology [Practical Diagnosis of Hematologic Disorders, 4th ed. (2006)]. The new reference ranges are not expected to significantly shift the percentage of patients flagged as having abnormal results; however, a shift of up to 5% in the number of patients with abnormal results for selected components such as hemoglobin may be seen.
Over the next 3 months, ESCL will make additional changes to CBC reporting that are aligned with accepted laboratory standards. These include the following changes (implemented in the order listed):
- Reporting absolute WBC counts (absolute neutrophil count, absolute lymphocyte count, etc.) with respective reference ranges.
Under most circumstances, clinically relevant WBC elevations and declines are defined by absolute counts, not by relative percentages. Current recommendations for the College of American Pathologists (CAP) and the Clinical and Laboratory Standards Institute (CLSI) recommend that absolute counts be the preferred method for reporting the WBC differential.
- Discontinuing reporting of reference ranges for relative (%) WBC differential counts as they are not the recommended method of reporting.
For questions related to CBC reference range changes, please contact your ESCL account representative.
East Side Clinical Laboratory is Testing for SARS-Cov-2/COVID-19
March 16, 2020
East Side Clinical Laboratory, Inc. is pleased to announce that testing for SARS-Cov-2/COVID-19 is available as of March 10th, 2020.
Due to strong market demand and limited supply nationwide, priorities will be given to high risk patients in accordance to CDC guidelines and endemic regions and clusters.
Test Information:
Test Code: COVIDHR - 17100
Test Name: SARS-COV-2 (COVID-19) BY RT-PCR, HIGH RISK
Ordering Recommendations:
Ordering provider to determine patient risk level based on CDC Guidelines and clinical judgement.
• Sonic has created an orderable code to triage patients, prioritize testing and coordinate with public health authorities:
o SARS-COV-2 (COVID-19) - High Risk: Patients who are symptomatic, exhibiting respiratory illness (cough, fever, and dyspnea) AND have traveled to a COVID-19 endemic area, have been in close contact with a known COVID-19 patient or are otherwise considered High Risk exposure from an epidemiological perspective.
• Criteria to Guide Evaluation of PUI (Persons Under Investigation) for COVID-19: The CDC currently states Clinicians should use their judgment to determine if a patient has signs and symptoms compatible with COVID-19 and whether the patient should be tested. Decisions on which patients receive testing should be based on the local epidemiology of COVID-19, as well as the clinical course of illness.
• Clinicians are strongly encouraged to test for other causes of respiratory illness, including infections such as influenza.
• In accordance with public health guidance, clinicians should prioritize testing to following groups:
1. Hospitalized patients who have signs and symptoms compatible with COVID-19 in order to inform decisions related to infection control.
2. Other symptomatic individuals, such as older adults and individuals with chronic medical conditions and/or an immunocompromised state that may put them at higher risk for poor outcomes (e.g., diabetes, heart disease, receiving immunosuppressive medications, chronic lung disease, chronic kidney disease).
3. Any person, including healthcare personnel, who within 14 days of symptom onset had close contact with a suspected or laboratory-confirmed COVID-19 patient, or who had a history of travel from affected geographic areas.
Specimen Requirements:
Sample Type:
• Upper Respiratory Tract: Nasopharyngeal Swab
Container Type: *Prefer dedicated collection device
• Nasopharyngeal swab: Viral or Universal Transport Media (VTM, UTM, VCM, UTM-RT, or M4) with Swab(s).
Must be ordered independent of other testing and include CDC PUI Form.
Handling Instructions:
Nasopharyngeal Swab: Insert a swab into the nostril parallel to the palate. Leave the swab in place for a few seconds to absorb secretions. Place swab immediately into sterile tube containing 2- 3 mL of viral transport media.
Transport: Critical Refrigerated
Unsuitable Specimens: Ambient specimens. Swabs not in viral transport media. Calcium alginate swabs. Swabs with wooden shafts. eSwab, any device refrigerated more than 72 hours post collection.
Testing Capacity:
Testing is performed seven days a week.
• Expected TAT is 1-3 days.
o TAT may vary with changes in capacity and market demands.
CPT Codes: U0001 – Real-Time Polymerase Chain Reaction (RT-PCR)
LOINC Codes: SARS-CoV-2 Interpretation (94306-8), Naso and Oro SARS-CoV-2 (94316-7), Nasopharyngeal SARS-CoV-2 (94316-7)
Download PDF
Sonic Healthcare USA Announces Testing Availability for COVID-19
Austin, Texas – March 13th, 2020
Sonic Healthcare USA has worked closely with its scientific partners and medical and operational leadership to bring up testing to ensure timely availability for patients in all geographies served by our laboratories. "We are closely monitoring capacity and are coordinating with our clinical laboratories to distribute testing that mitigates high demands from endemic regions and clusters in the United States," said Jerry Hussong, MD, MBA, the Chief Executive Officer of Sonic Healthcare USA.
Sonic has taken comprehensive measures to expand testing capacity, while maintaining high quality testing and meeting required turnaround times. As a result, Sonic is bringing up multiple testing platforms and methodologies.
Dr. Hussong added, "In response to this public health emergency, Sonic Healthcare USA pledges to do its part and is actively monitoring the situation. Our commitment is to ensure testing is available as this pandemic evolves." The well-being of our patients and employees is our highest priority, and we continue to be committed to the medical communities we serve.
About Sonic Healthcare USA
Sonic Healthcare USA is a subsidiary of Sonic Healthcare Limited, one of the world's largest medical diagnostic companies, providing laboratory services to medical practitioners, hospitals and community health services, with operations in eight countries, on three continents and providing care to over 100 million patients each year. Sonic Healthcare USA is a leading provider of state-of-the-art laboratory services and pathology practices throughout the USA with nine operating divisions and nearly eight thousand US-based employees. Sonic Healthcare USA utilizes a federated business model that emphasizes medical leadership and community-based testing services to provide outstanding quality and service to the doctors and patients that they serve. For more information, visit the Sonic Healthcare website at www.sonichealthcareusa.com.
| Sonic Healthcare USA Media Contact Chief Executive Officer |
Sonic Healthcare USA and other Clinical Laboratory Executives Met with the White House's Coronavirus Taskforce
Austin, Texas – March 6th, 2020
This week, Academic and Commercial Reference Laboratory Executives met with Vice President, Mike Pence and members of the White House's Coronavirus Task Force to collectively discuss diagnostic testing availability for COVID-19.
Jerry Hussong, MD, MBA, the Chief Executive Officer of Sonic Healthcare USA (Sonic), was among those who met on behalf of the American Clinical Laboratory Association (ACLA) with key government officials, the CDC, and the FDA. At the meeting, key stakeholders discussed the role of state and local public health and commercial laboratories, hospitals, and academic medical centers to increase access to testing for the Coronavirus. "Our main goal, as a commercial laboratory, is to ensure those patients in need or at high-risk can access testing. By working together, we can accelerate those efforts with a coordinated and comprehensive approach," said Dr. Hussong.
Dr. Hussong added, "Sonic Healthcare is working with its Medical, Scientific and Operational Leadership to make patient testing available through its network of commercial laboratories in the United States and will follow the FDA guidelines for Emergency Use Authorizations (EUAs)." The FDA has recently updated its policy, thus providing an expedited pathway for the availability of diagnostics for COVID-19.
Sonic Healthcare will continue to lead and collaborate with colleagues, government officials, and our local medical communities in response to this immediate public health crisis.
Read the ACLA Statement on COVID-19 Testing
About Sonic Healthcare USA
Sonic Healthcare USA is a subsidiary of Sonic Healthcare Limited, one of the world's largest medical diagnostic companies, providing laboratory services to medical practitioners, hospitals and community health services, with operations in eight countries, on three continents and providing care to over 100 million patients each year. Sonic Healthcare USA is a leading provider of state-of-the-art laboratory services and pathology practices throughout the USA with nine operating divisions and nearly eight thousand US-based employees. Sonic Healthcare USA utilizes a federated business model that emphasizes medical leadership and community-based testing services to provide outstanding quality and service to the doctors and patients that they serve. For more information, visit the Sonic Healthcare website at www.sonichealthcareusa.com.
| Sonic Healthcare USA Media Contact Chief Executive Officer |
2019
Discontinuation of 48-hour Urine Culture Incubation
Issued December 10, 2019: The previous protocol for urine cultures being processed by East Side Clinical Laboratory was to re-incubate for an additional 24 hours if no growth was observed initially on day 1. Current understanding of urinary pathogens has now rendered this re-incubation unnecessary. Most causative agents of UTI’s can be identified by routine urine culture, which has been published as not benefitting from 48 hours of incubation. East Side Clinical Laboratory will continue to re-incubate cultures from catheterized specimens, children 10 years of age or younger, or if a clinician suspects a fastidious pathogen that requires additional incubation.
Our goal at East Side Clinical Laboratory is to ensure that patients receive results in a timely manner without compromising testing. By reporting urine cultures in 24 hours, we ensure results are communicated timely to guide treatment decisions. Additional incubation on routine urine cultures will not be performed unless specifically requested.
For more information regarding the change in protocol, please contact your ESCL representative or the ESCL Microbiology Supervisor (Kelly Messier, 401-455-8427).
Thank you for allowing us to continue to provide the highest quality laboratory testing services to you and your patients.
REFERENCES:
- Wilson ML, Gaido L. Laboratory Diagnosis of Urinary Tract Infections in Adult Patients. Clinical Infectious Diseases. 15 Apr 2004,38(8):1150-1158. DOI 10.1086/383029.
- Shoskes D. The American Urological Association Educational Review Manual in Urology. Third Edition. Chapter 23: Urinary Tract Infections. 2011.
- Ulett KB, Benjamin Jr WH, Zhuo F, et al. Diversity of Group B Streptococcus Serotypes Causing Urinary Tract Infection in Adults. ASM Journal of Clinical Microbiology. July 2009,47(7):2055-2060. DOI 10.1128/JCM.00154-09
The FDA Warns: BIOTIN May Interfere with Laboratory Testing*
- Patients on high dose supplements of biotin (minimum 5-10 mg/day), such as hair/nail/ skin supplements, should refrain from taking them in the 24 hours prior to laboratory test.
- Supplements may or may not prominently indicate they contain biotin.
- Patients taking prescribed megadose regimens (>150 mg biotin, usually prescribed for multiple sclerosis) should refrain from taking the supplements for 2-3 days prior to laboratory testing, after consulting with their physician.
THE LEVEL OF BIOTIN TYPICALLY FOUND IN MULTIVITAMIN TABLETS DOES NOT INTERFERE WITH LABORATORY TESTING. *Patients and physicians should review any lab test result that does not match the clinical presentation and consider if biotin interference is present.
Discontinuation of H. pylori Serology Testing
Effective November 4, 2019, East Side Clinical Laboratory will no longer offer Helicobacter pylori (H. pylori) serology testing based upon changes in H. pylori testing guidelines of the American College of Gastroenterology and the American Gastroenterology Association. The discontinued antibody tests below will be inactivated in our compendium and will not be orderable. Serological antibody results have poor positive predictive value (PPV) and are no longer considered clinically useful. The alternative noninvasive testing methods demonstrate higher clinical utility, sensitivity, and specificity.
A positive H. pylori IgG, IgA, or IgM result cannot be used to predict the presence of active disease. Since antibodies to H. pylori remain detectable for years following resolution of infection, serologic testing cannot be used to distinguish active from past infection or to document eradication of the organism following successful treatment.
Many insurance companies are no longer reimbursing patients for whom H. pylori serologic testing was performed, as they consider such testing “not medically necessary”. As an alternative, providers are encouraged to collect samples for Urea Breath Test or Stool Antigen Test. Both assays are FDA cleared for use to detect H. pylori infection, and as a test of eradication. Clients should order collection kits as needed with instructions for use.
Discontinued Test With Order Codes:
| Order Code | Test |
|---|---|
| 4138 | H. pylori IgG, IgA, IgM Antibodies |
| 4091 | H. pylori IgG Antibody |
| 4140 | H. pylori IgA Antibody |
| 4141 | H. pylori IgM Antibody |
Recommended Tests With Order Codes
| Order Code | Test |
|---|---|
| 1832 | H. pylori Antigen, Feces |
| 5173 | H. pylori Breath Test |
If you have any questions please call East Side Clinical Laboratory at 800.455.8440 or contact your East Side Clinical Laboratory representative.
APTIMA Multitest Orange Swab
Effective September 15, 2019 East Side Clinical Laboratories will offer a new collection device, the APTIMA Multitest Orange Swab for Bacterial Vaginosis, Candida, Gonorrhea (GC), Chlamydia (CT) and Trichomonas (Trich) Amplification Testing. This swab can be used in place of the white label APTIMA swab (with the exception of endocervical and male urethral samples).
APTIMA Multitest Orange Swab Utilization
| Clinician Collected | ESCL Test Code |
|---|---|
| GC Vaginal | 4180 |
| CT Vaginal | 4179 |
| Trichomonas Vaginal | 5650 |
| GC/CT Vaginal | 4181 |
| GC/CT Trich Vaginal | 5780 |
| GC/CT Oral | 5740 |
| GC/CT Anal | 5698 |
| Bacterial Vaginosis Panel | 15700 |
| Bacterial Vaginosis, Candida, Trichomonas | 16201 |
| Bacterial Vaginosis, Chlamydia/GC | 16202 |
| Bacterial Vaginosis, Candida, Chlamydia/GC, Trichomonas | 16203 |
| Candida by PCR | 15800 |
| Mycoplasma/Ureaplasma PCR | 16770 |
| HSV I/II, VZV by PCR | 13322 |
| HSV I/II by PCR | 5367 |
Specimen Information
| Turn Around Time | Analytic time 2-5 days after receipt |
|---|---|
| Transportation | Room temperature (15-25°C) or refrigerated (2-8°C) |
| Stability | After inoculation, sample stable in collection device for 60 days |
| Rejection Criteria | Empty collection devices, unlabeled samples and leaking samples |
Must Call Critical List
| Value stated and less than | Value stated and greater than | ||
|---|---|---|---|
| Amylase |
|
| 400 |
| Calcium |
| 6 | 13 |
| CKMB
|
|
|
7.8 4.4 |
| CO2 |
| 10 | 40 |
| CPK |
|
| 400 |
| Creatinine |
|
| 5 |
| CSF (Cerebral Spinal Fluid) | All Results |
|
|
| Cultures
| All Positives |
|
|
| D-Dimer |
|
| 0.51 |
| Glucose |
| 50 | 400 |
| Hemoglobin |
| 7 |
|
| Malaria/Babesia on peripheral smear | Call/page all positive or suspicious results |
|
|
| Platelet |
| 20,000 | 1,000,000 |
| Potassium |
| 3 | 6.5 |
| Potassium (heparin) |
| 3 | 6.5 |
| PT/INR |
|
| 5 INR |
| PTT |
|
| 70 |
| Sodium |
| 120 | 160 |
| Therapeutic Drugs |
|
|
|
|
|
| 2.1 |
|
|
| 30 |
|
|
| 2.0 |
|
|
| 50 |
| Total Bilirubin |
|
| 5 |
| Troponin | Call/page if abnormal |
|
|
| WBC-Adult/child |
| 1.5 | 50.0 |
2018
HPV Low Risk Discontinuation
Effective January 1, 2019, East Side Clinical Laboratory (ESCL) will discontinue offering combined testing for high and low risk Human Papillomavirus (HPV). Low Risk HPV will no longer be performed. This change is due to the implementation of newer analytic methods and testing algorithms.
The following tests/order codes will be deleted with this update:
| Test Name | Test Code |
|---|---|
| PAP and HPV (high and low risk) | 4176 |
| PAP with GC/Chlamydia | 4178 |
| HPV (high and low risk) | 4196 |
| PAP smear with HPV Reflex for Ascus/Agus | 4182 |
HPV low risk will be removed from the following test codes with this update:
| Test Name | Test Code |
|---|---|
| PAP with HPV reflex Ascus/Agus and GC Chlamydia | 4184 |
| PAP with HPV reflex for all abnormalities and GC/Chlamydia | 4193 |
As an alternative, ESCL offers Roche high risk HPV testing with genotyping information. Amongst high risk HPV infections, genotypes 16 and 18 are most strongly associated with cervical cancer and severe Dysplasia/CIN3.
For high risk testing with genotype information, we offer the following:
| Test Name | Code |
|---|---|
| HPV Genotypes 16 and 18 | 9848 |
| HPV Genotypes 16 and 18/45 | 15670 |
| HPV High Risk Genotype 16/18 with reflex PAP | 10075 |
| HPV mRNA E6/E7 | 4174 |
| HPV mRNA Genotpyes 16, 18/45 | 16565 |
| HPV mRNA with reflex to Genotypes 16, 18/45 | 16564 |
High risk HPV:
- Co-testing is endorsed by the American Congress of Obstetricians and Gynecologists (ACOG) and other authorities for woman 30-65 years of age
- Algorithms adopted by the American Society for Colposcopy and Cervical Pathology (ASCCP) recommend high risk HPV genotyping for types 16 or 18 as an immediate alternative to a return visit for a pelvic examination and repeat co-testing for those patients found to be high risk HPV positive and PAP negative(see algorithms at www.asccp.org/Guidelines)
- Genotyping for 16 and 18 can be used to refine management of women 21-29 years of age in certain clinical scenarios
For questions related to HPV testing options, please contact your ESCL account representative.
Quantiferon Gold 4 Tube Testing
East Side Clinical Laboratory is pleased to announce the introduction of the QuantiFERON-TB Gold Plus assay which offers optimized analysis with tuberculosis (TB)-specific antigens that elicit both CD4+ and CD8+ T-cell responses. The new 4-tube collection set enables a more comprehensive assessment of cell-mediated immune responses to TB infection
Clients should begin to order QFT - 4 tube kits but may continue to send the 3-tube collection kits until January 1, 2019 when the 3-tube version will be discontinued.
The 4-tube assay requires the same pre-analytical processing steps as the current 3 tube QuantiFERON Gold assay. Immediately after filling the tubes with 1ml blood, shake 10 times just firmly enough to coat the inner surface of the tubes without disrupting the gel.
Please use test code 9884 when ordering the 4-tube assay. Tubes should remain at room temperature for same day transport to ESCL
For questions related to TB GOLD Plus testing, please contact your ESCL account representative.
Thank you for choosing East Side Clinical Laboratory




 Aptima® Multitest (Orange Aptima) and Aptima® Unisex (Blue Aptima)
Aptima® Multitest (Orange Aptima) and Aptima® Unisex (Blue Aptima)